Youngkwang Lee, Ph.D.

Pronouns: Youngkwang
Assistant Professor of Biophysical and Analytical Chemistry
Department of Chemistry and Biochemistry
Analytical Chemistry
College of Sciences
SDSU
Primary Email: [email protected]
Phone/Fax
Primary Phone: 42422
Building/Location
Engineering and Interdisciplinary Sciences Complex - 17
5500 Campanile Dr
San Diego,
CA
92182
Bio
My lab explores the molecular underpinnings of biochemical reaction systems using advanced imaging and spectroscopic techniques. Our current research focuses on uncovering the structural and functional dynamics of proteins involved in cell signaling pathways.
Areas of Specialization
Biophysical Chemistry, Analytical Chemistry
Research
Research Focus
The Youngkwang Lee group applies advanced imaging and spectroscopic techniques to solve important problems in cell signaling and develop novel biosensing platforms. With an emphasis on optical analyses, we use research methods from multiple disciplines — biophysics, biochemistry, materials science and nanotechnology. We integrate these tools into our research program to quantitatively analyze biomolecular reactions at single-molecule resolution. The research program in the lab has two main areas of interest described below.
Current Research Area
Single-molecule analysis of signaling reactions
Many of cellular processes involve a series of biochemical reactions that take place on membranes. The importance of membrane reactions is underscored by the fact that more than 50% of current drugs target membrane proteins and many of cytosolic proteins are fully functional only within membrane environments. Reactions on membrane surfaces are intrinsically different from those in solution.
Proteins experience enhanced local concentrations, orientational and conformational change, restricted diffusion, and physical assembly with their interacting partners; as a result, many emergent properties arise at the membrane. However, a relative lack of methodology for quantitatively analyzing membrane processes effectively hinders our understanding of cell signaling.
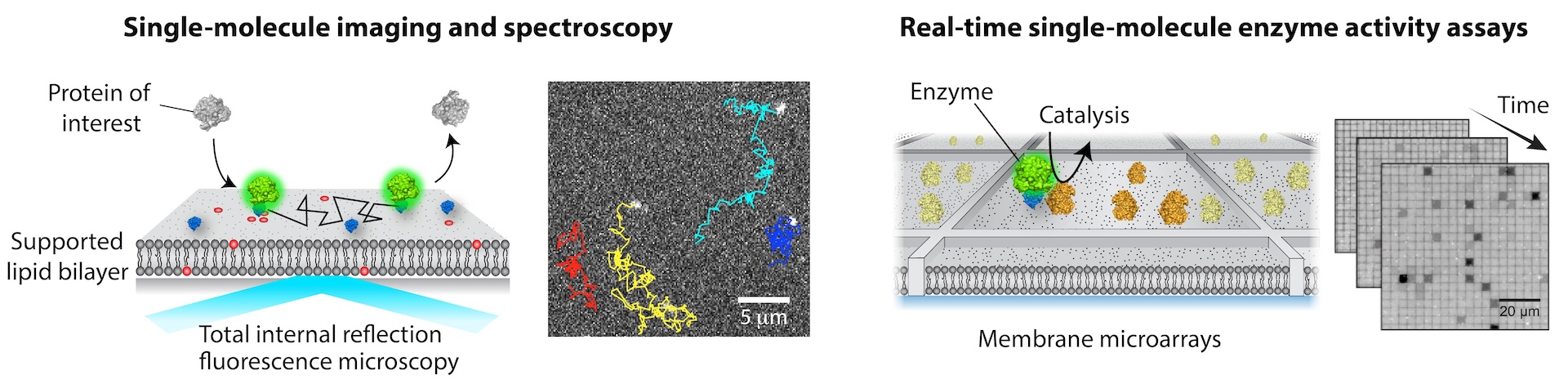
A major focus in the lab is to solve this challenge by bringing in new technologies. A key idea is to reconstitute signaling reactions on a supported lipid bilayer (SLB) in a highly controlled manner. A suite of cutting-edge single-molecule fluorescence imaging and spectroscopic techniques are employed to quantitatively elucidate detailed molecular mechanisms. Remarkably, single-molecule readout is achievable even with direct cell extract. Our approach is generally applicable to other systems, beyond membrane signaling.
Overall, we apply chemical and physical principles to understand complex biological phenomena, seeking to leverage our discovery to develop better therapeutic strategies. The current projects include molecular assembly and activation of small GTPases and Raf kinases at the membrane.
Functional nanostructure/biomolecule hybrids for novel bioassays
Quantification of extremely low-abundance biomolecules (e.g. nucleic acids and protein) is of tremendous importance for applications such as early diagnosis, adjuvant therapy, food safety monitoring, and biological warfare defense. We aim to develop novel optical sensors by synthesizing functional nanostructure/biomolecule hybrids. Our primary synthetic building blocks include plasmonic nanoparticles, artificial cell membranes, DNA nanomachines and recombinant proteins. The ultimate detection sensitivity of the hybrid sensing system is at the single-molecule/particle level.
Students working in my lab will be trained to be skillful at a variety of cutting-edge imaging and spectroscopic techniques including single-molecule total internal fluorescence microscopy (TIRF), dark-field microscopy, fluorescence correlation spectroscopy (FCS), single-molecule fluorescence resonance energy transfer (FRET), and time-correlated single photon counting (TCSPC). Additionally, they will receive an interdisciplinary training to build a broad background in nanoparticle synthesis, surface chemistry, nano/micro fabrication, high-throughput data analysis, and protein purifications.
